 Medical Devices
Medical Devices
SurePath™ Gynaecological Liquid-Based Cytology System
The SurePath™ Liquid-Based Cytology System is the only FDA-certified LBC method clinically proven to reduce unsatisfactory specimen rates by 58%, offering the highest cervical precursor lesion detection sensitivity among all commercially available LBC platforms.
Read More Medical Devices
Medical Devices
Affirm VPⅢ Chip Assay in Vaginal Secretion Routine Testing
A multicentre clinical study demonstrates high concordance between the Affirm VPⅢ Chip Assay and gold-standard reference methods for the simultaneous detection of Bacterial Vaginosis, Vulvovaginal Candidiasis and Trichomonas Vaginalis in routine vaginal secretion testing.
Read More Medical Devices
Medical Devices
Affirm™ VPIII Vaginal Infection Microbial Detection System
The Affirm™ VPIII system uses industry-first DNA probe technology to detect three major vaginal infections — Bacterial Vaginosis, Vulvovaginal Candidiasis and Trichomonas Vaginalis — simultaneously from a single specimen in a fully automated, operator-independent workflow endorsed by the US CDC.
Read More Medical Devices
Medical Devices
Viper LT™ System and Onclarity™ HPV Assay
The Viper LT™ System combined with the Onclarity™ HPV Assay delivers comprehensive individual genotyping results for all 14 high-risk HPV types from a single specimen — targeting E6/E7 oncogenes for maximised sensitivity and enabling personalised cervical cancer risk stratification in a fully automated walk-away platform.
Read More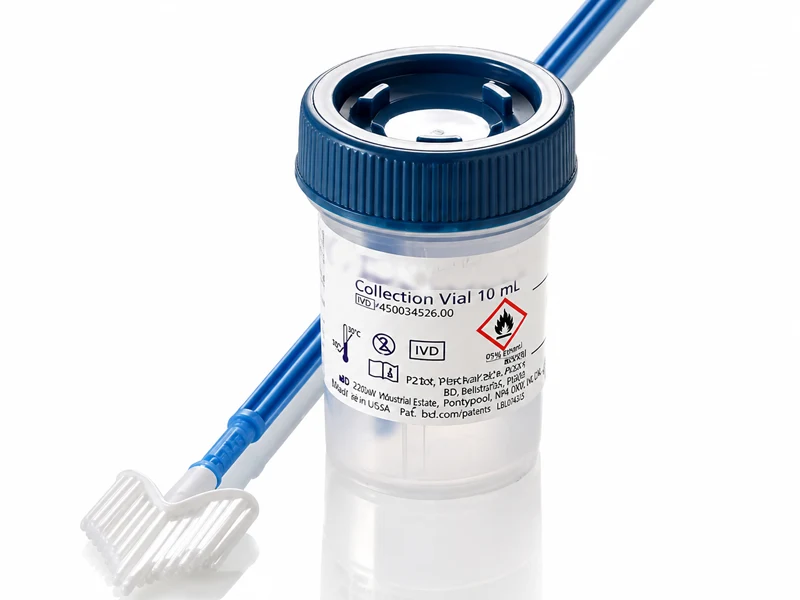 Industry Trends
Industry Trends
Clinical Significance of Non-Gynaecological Liquid-Based Cytology
LBC technology extends well beyond cervical cancer screening — with proven applications in sputum, urine, pleural and peritoneal effusion cytology that significantly improve lesion detection rates over conventional direct smear methods, opening a substantially expanded market opportunity for liquid-based cytology platforms.
Read More