Medical Devices · Cervical Cancer Screening · FDA & NMPA Certified


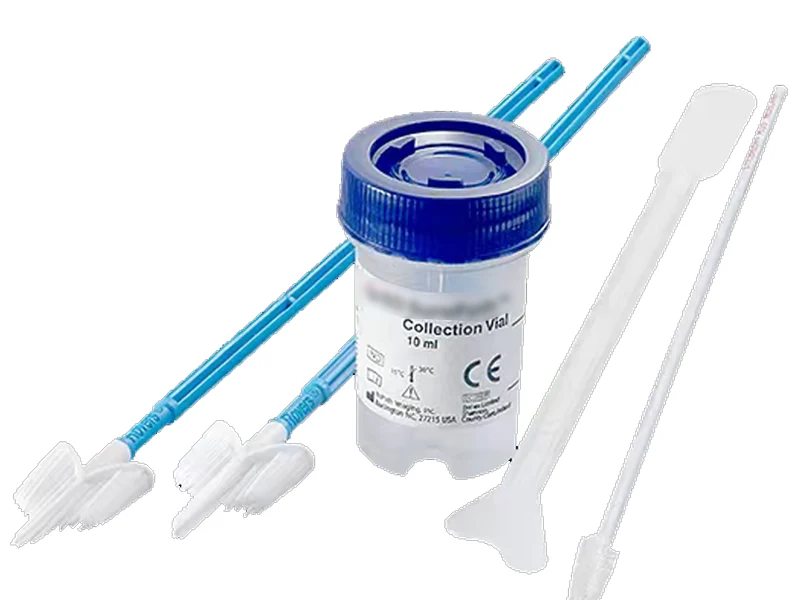
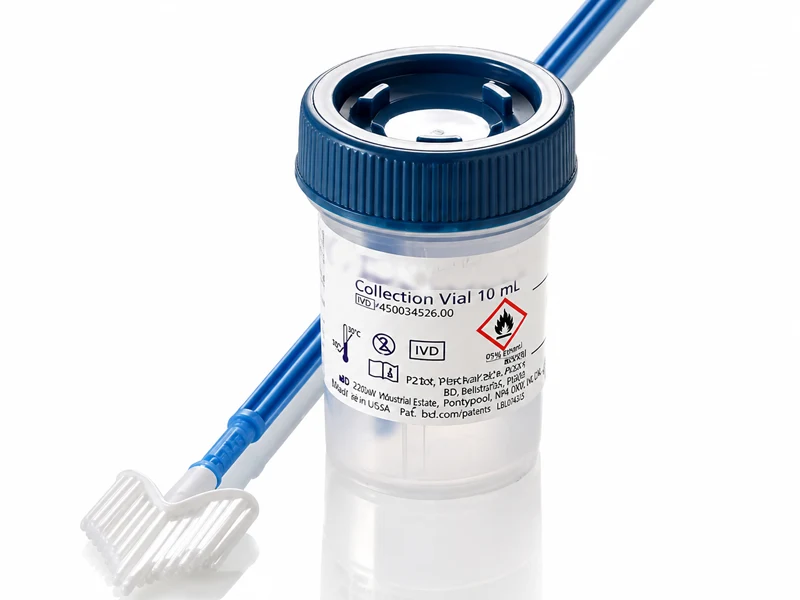
The SurePath™ LBC system uses a proprietary sample collection method and patented centrifugal sedimentation technology — the third-generation cytology testing method. It delivers automated, standardised, high-throughput slide preparation with industry-leading specimen adequacy rates and the highest FDA-certified cancer precursor detection rate compared to conventional Pap smears.
SurePath™ liquid-based cytology is the world’s most clinically validated cervical screening technology, backed by over two decades of large-scale FDA clinical evidence. Its unique sedimentation-based slide preparation preserves the three-dimensional cellular structure, producing a clear, uniform monolayer free from obscuring blood, mucus and inflammatory cells.
The PrepStain™ Slide Processor automates the entire workflow — from sample pre-treatment and cell enrichment to slide preparation and Papanicolaou staining — completing a batch of 24 slides in just 40 minutes. Each slide uses an independent reagent cartridge to eliminate cross-contamination, and the closed waste-liquid system reduces fume exposure in the laboratory.

| Model | PrepStain™ Slide Processor |
|---|---|
| Dimensions | 770 × 580 × 650 mm (W × D × H) |
| Weight | 65 kg |
| Throughput | 1–24 slides per batch · 40 min per batch of 24 |
| Slide Preparation Method | Sedimentation (preserves 3D cell structure) |
| Staining Method | Independent single-slide drop staining (no cross-contamination) |
| Applications | Gynaecological LBC · Non-gynaecological cytology · Immunocytochemistry (dual / single stain) |
| Reagent System | Independent cartridge per slide; closed waste-liquid system |
| Operating Temperature | 25–30 °C |
| Regulatory Status | FDA 510(k) cleared · NMPA registered · CE IVD |
| Compatible Media | SurePath™ preservative fluid |